
Seguridad: evaluación de la seguridad de dosis crecientes de CBD en perros sanos
Investigación preliminar de la seguridad de dosis crecientes de cannabinoides en perros sanos
Vaughn, J. Kulpa y L. Paulionis
2020
CONCLUSIONES CLAVE:
- La administración de aceite de CBD tuvo poco efecto sobre la ingesta de alimentos y la actividad física
- No hubo diferencia en el perfil de efectos adversos del aceite de CBD en comparación con su grupo de control
- La administración de aceite de CBD hasta una dosis de 62 mg/kg por día solo produjo efectos secundarios leves y estos efectos secundarios también se detectaron en el grupo de placebo correspondiente
- No se informaron efectos adversos moderados o clínicamente significativos después de la administración de aceite de CBD hasta 62 mg/kg al día.
- El estudio proporciona datos novedosos que separan la seguridad y la tolerabilidad del aumento de dosis de las formulaciones de aceite predominantes en CBD, THC o CBD + THC (1,5:1)
- Hubo una clara distinción en el perfil de seguridad del aceite de CBD en comparación con el aceite de THC o CBD+THC, siendo el aceite de CBD el mejor tolerado
- El estudio respalda el perfil seguro y tolerable del aceite de CBD incluso cuando se administra hasta 62 mg/kg al día, lo que se considera una dosis mucho más alta de lo que se esperaría para uso clínico.
- Las dosis normalmente recomendadas oscilan entre 0,5 y 8 mg/kg.
OBJETIVOS E HIPÓTESIS → Dado el creciente interés y la evidencia de las propiedades terapéuticas del CBD en animales de compañía, existe la necesidad de evaluar el perfil de seguridad del CBD. El estudio se propuso evaluar la seguridad y la tolerabilidad de dosis crecientes de tres formulaciones de aceite de cannabis, que contienen predominantemente CBD, THC o CBD y THC (1,5:1) frente a placebo en perros. Un objetivo secundario fue determinar los niveles sanguíneos de CBD, THC y sus metabolitos a dosis más altas de CBD y THC.
MÉTODOS → El estudio fue un estudio aleatorizado, controlado con placebo, ciego y paralelo que inscribió a 20 perros sanos. Los perros fueron asignados al azar a uno de los cinco grupos de tratamiento: aceite con predominio de CBD, aceite con predominio de THC, aceite con predominio de CBD/THC (1,5:1), placebo de aceite de girasol (SF), placebo de aceite de MCT (triglicéridos de cadena media). Se planificaron administraciones de hasta 10 dosis escalonadas de los aceites, con al menos 3 días de separación entre dosis (las dosis máximas diarias administradas fueron: 62 mg/kg de CBD, 49 mg/kg de THC y 12 mg/kg de CBD + 8 mg /kg de THC). Se usaron dos aceites de placebo diferentes ya que los aceites de cannabinoides incluían aceite SF o MCT como solventes. Veterinarios y técnicos veterinarios experimentados realizaron las observaciones clínicas y las evaluaciones físicas, y también se midieron los hemogramas completos, la química clínica y los cannabinoides plasmáticos para evaluar la seguridad, la tolerabilidad y la aparición de eventos adversos (AA).
RESULTADOS → De los aceites probados, el aceite de CBD tuvo el menor efecto sobre la ingesta de alimentos y la actividad física. Con respecto a la interrupción del estudio, 1 perro en el grupo de aceite de THC y 2 en el grupo de CBD/THC fueron interrumpidos del estudio debido a efectos adversos (AA) graves. Ningún perro fue descontinuado de los grupos de aceite de CBD o aceite de placebo como resultado de AE. Se informaron efectos secundarios en los 5 grupos, y de los 505 EA que se registraron en total, 104 ocurrieron en los grupos de placebo, 80 en el grupo de CBD, 206 en el grupo de THC y 115 en el grupo de CBD/THC. El 94,9% de estos efectos adversos fueron leves. Centrándose únicamente en el grupo de aceite de CBD, vale la pena destacar que el número y el perfil de los EA fue similar al grupo de aceite de placebo de MTC. Además, no hubo efectos adversos moderados en el grupo de aceite de CBD en ninguna de las dosis probadas, mientras que en su grupo de placebo correspondiente, 2 perros sufrieron efectos adversos leves. Además, no hubo efectos secundarios graves o médicamente significativos en el grupo de aceite de CBD en ninguna de las dosis probadas.
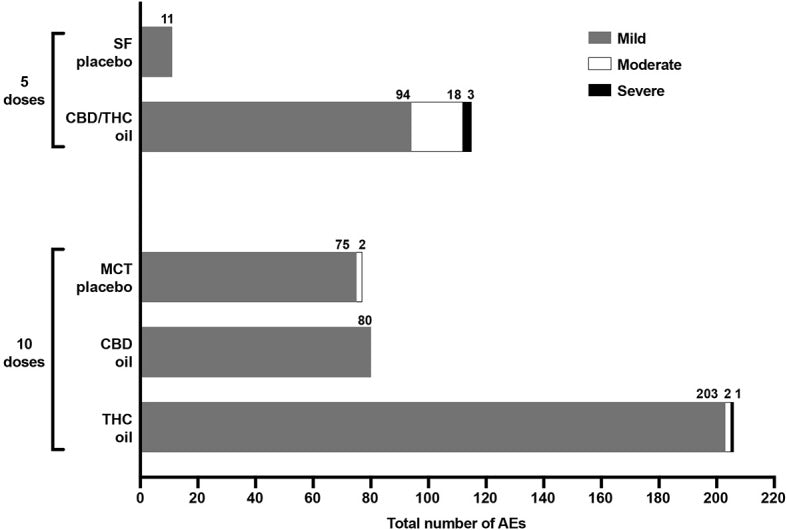
Figura 1: número total y gravedad de los AA experimentados en 5 dosis crecientes de placebo de aceite SF y aceite de CBD/THC y 10 dosis crecientes de placebo de aceite MCT, aceite de CBD y aceite de THC

Figura 2: Número total de EA leves por categoría anatómica. AE leves en 10 dosis de placebo de aceite MCT, aceite de CBD o aceite de THC.
Se produjeron AE moderados en el 40 % de los sujetos en tres grupos: 2 perros en el grupo de aceite MCT, 2 perros en el grupo de aceite THC y 4 perros en el grupo de aceite CBD/THC. Se produjeron efectos adversos graves en el 15 % de los sujetos del grupo de aceite de THC y aceite de CBD/THC. Con respecto a los análisis de sangre y químicos, el único cambio determinado fue un aumento en ALP en un perro en el grupo de aceite de CBD (cuando se administró la décima dosis) y en un perro en el grupo de aceite de CBD/THC (quinta dosis). Estos cambios no se consideraron clínicamente significativos y cuando estos niveles se volvieron a medir 7 días después de la dosis final, hubo una tendencia a la baja. Los niveles plasmáticos de CBD, THC y sus metabolitos fueron muy variables entre los perros del mismo grupo de tratamiento que recibieron la novena dosis de aceite de CBD o de aceite de THC. Después de la novena dosis del aceite de CBD, los niveles máximos de CBD en plasma alcanzados en los cuatro perros del grupo de aceite de CBD fueron comparables al rango de niveles de CBD en plasma alcanzado en nueve perros después de dosis diarias repetidas de CBD (2,5 mg/kg dos veces al día) durante 12 semanas . En base a estos resultados, parece que un efecto de primer paso a través del hígado no eliminó la disponibilidad sistémica de CBD luego de su ingestión oral. Además, el hecho de que se observaran niveles cuantificables de CBD una semana después de la exposición a la dosis también es interesante, dado que se ha informado que el CBD tiene una eliminación relativamente rápida en perros.
En conclusión, los hallazgos proporcionados por el estudio respaldan el perfil de seguridad del CBD y respaldan su potencial como opción de tratamiento en medicina veterinaria.
